Down Syndrome Abstract
of the Month: June 2000

Mechanisms of neuronal death in Down's syndrome
Nagy Zs.
J Neural Transm Suppl 1999;57:233-45
Departments of Neuropathology and Pharmacology, Univ of Oxford, UK.
Summary:
There is growing evidence that neuronal death in Down's syndrome is due to apoptotic mechanisms. The phenomena, however, that trigger and regulate programmed cell death in the Down's syndrome-related neurodegeneration are still much debated.In vitro evidence has suggested that the main factor responsible for the neuronal death in this condition is the accumulation of beta-amyloid, due to the overexpression of its precursor protein. Another hypothesis argues for the importance of reactive oxygen species in neuronal death. However, the in vivo findings do not entirely support either theories.
We propose that neuronal apoptosis, as well as the formation of Alzheimer-type pathology, in Down's syndrome is due to an aberrant re-entry of neurons into the cell division cycle. Due to the simultaneous overexpression of conflicting cell cycle regulatory signals, the mitogenic amyloid precursor and the differentiation factor S100, the cell cycle is abandoned. Subsequently the cell cycle arrest may lead to either the formation of Alzheimer-related pathology or to apoptotic cell death.
My comments:
This is a very well written paper. Dr Nagy, who has done previous work on this topic, starts by reviewing the information known to date on Down syndrome brain pathology. I'll try to hit the high points.
The brain pathology most associated with DS consists of decreased cell numbers, altered neuronal differentiation, altered synaptic connectivity and the development of amyloid plaques (AP) and neurofibrillary tangles (NT). The main constituent of the plaques is the protein beta-amyloid, which is produced when a bigger protein, the amyloid precursor protein (APP) is cleaved by an enzyme. The gene making APP is located on the 21st chromosome and is thought to be overexpressed in DS. The amyloid proteins tend to cluster in regions in Alzheimer's, but are fairly diffusely located in the brains of adults with DS.
Apoptosis is programmed cell death. In "normal" brains, this is used to remove redundant neurons as development occurs, thus setting pathways. Proteins called "caspases," which are present in every cell, are the "executors" of the nucleus which lead to the self-destruction of the cell. The proteins that block the apoptosis are the Bcl-2 family of proteins. So whether a cell lives or undergoes apoptosis (I don't think "apoptoses" is a verb) depends upon which group of proteins outnumber the other group.
Previous studies have documented the loss of neurons over the span of life of a person with DS, and studies in the last decade have implicated apoptosis as the cause. Now, what causes the apoptosis? Here are the current hypotheses:
1. Beta-amyloid toxicity: some studies indicate that the amyloid deposition disrupts the synaptic integrity, meaning that the neurons don't communicate well with each other anymore. Other studies indicate that the amyloid may be directly toxic to neurons in culture plates, as well as making neurons more sensitive to neurotoxic insults (alcohol, Adam Sandler, etc). The problem is that amyloid presence itself doesn't necessarily relate to severity of dementia, and amyloid doesn't relate well to NF which does correlate well to dementia in non-DS adults with Alzheimer's.
2. Free-radical theory: Since Superoxide Dismutase is overexpressed in DS, it's possible that oxidative stress may be involved in neuronal death in DS. While in vitro studies have shown increased sensitivity to oxidative stress of DS neurons, two post-mortem studies in DS have shown no evidence of a pathogenic role of SOD or reactive oxygen species.
3. Abortive mitosis theory: This is somewhat difficult to describe. By looking for proteins that are specific for certain phases of the cell cycle, researchers found that the neurofibrillary tangles were related to disruptions in cell cycle regulation. This author in a previous study went looking for signs of disruption in cell cycle regulation in DS a few years ago and found that neurons in the part of the brain called the hippocampus are not static but have the ability to re-enter the cell cycle, which is not supposed to happen. If these cells do re-enter the cell cycle, they appear to "stall" just before the mitosis phase. In some parts of the brain, this abortive mitosis phenomenon is followed by the appearance of tangles, but in the hippocampus of DS, cell death occurs instead. The author believes this genetically determined failure of cell cycle regulation is at the root of brain pathology in DS.
Here's the bottom line: the author proposes a "death cascade" for neurons in DS. Here's a diagram from her paper:
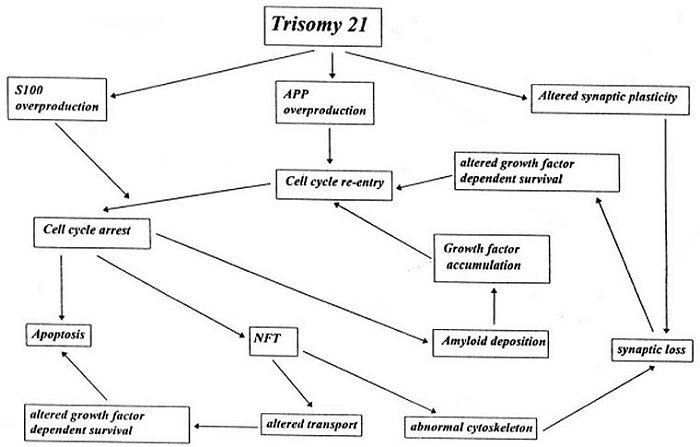
The third 21st chromosome causes altered synaptic plasticity (the ability of neurons to form new pathways), as well as overproduction of S100, SOD and APP.
The altered synaptic plasticity leads to synaptic loss which leads to altered survival of the neurons, dependent upon nerve growth factor, which leads to abortive cell mitosis. Abortive cell mitosis leads to amyloid deposition and apoptosis. S100 and APP overproduction leads to abortive cell mitosis, and apoptosis as well. SOD overproduction leads to increased sensitivity of the neurons to neurotoxins or just predisposes the neurons to apoptosis.
The author postulates, therefore, that this genetically determined failure of cell cycle regulation is at the root of all brain pathology in Down syndrome.